
The average atomic mass is a)12.5 b)12.1 c)6.05 d)6.1? tests, examples and also practice JEE tests. Carbon-14 is a natural radioactive isotope with a. The most common is stable carbon-12, followed by stable carbon-13. Carbons atomic radius is 77 picometers, silicons is 118 picometers, germaniums is 123. Carbon has the atomic number of 6 which means that all isotopes have the same proton number. The atomic radii of the carbon group elements tend to increase with increasing atomic number. For example, any atom that contains six protons is the element carbon and has the atomic number 6, regardless of how many neutrons or electrons it may have. Carbon-14, an isotope with a half-life of 5715 years, has been widely used to date such. In 1961 the International Union of Pure and Applied Chemistry adopted the isotope carbon-12 as the basis for atomic weights. All of these isotopes have the same atomic number but different mass numbers. Atomic Number: 6: Atomic Radius: 170 pm (Van der Waals) Atomic Symbol: C: Melting Point: 3550 ☌ (diamond) Atomic Weight: 12.01. The most common of these is carbon 12, 13, 14. The average atomic mass is a)12.5 b)12.1 c)6.05 d)6.1? theory, EduRev gives you anĪmple number of questions to practice A sample of carbon that contains 95% by mole carbon-12 and 5% by mole carbon-14 isotopes. Explanation: Carbon exists in several isotopes. The average atomic mass is a)12.5 b)12.1 c)6.05 d)6.1? has been provided alongside types of A sample of carbon that contains 95% by mole carbon-12 and 5% by mole carbon-14 isotopes. Carbon-12 is the common isotope, with carbon-13 as another stable isotope which makes up about 1. The average atomic mass is a)12.5 b)12.1 c)6.05 d)6.1?, a detailed solution for A sample of carbon that contains 95% by mole carbon-12 and 5% by mole carbon-14 isotopes. The element is determined by the atomic number 6. The number of electrons in a neutral atom is equal to the number of protons. Cosmic rays enter the earths atmosphere in large numbers every day and when one collides with an atom in the atmosphere, it can create a secondary cosmic ray. The number of protons in the nucleus of the atom is equal to the atomic number (Z). Besides giving the explanation ofĪ sample of carbon that contains 95% by mole carbon-12 and 5% by mole carbon-14 isotopes. The number of protons, neutrons, and electrons in an atom can be determined from a set of simple rules. Carbon 12 makes up for around 99 per cent of total carbon, while Carbon 14 makes up only one per cent. Carbon 12 has an atomic mass of 12,011 AMU, while Carbon 14 has an atomic mass of 14.0032420 u. Note: Carbon-14 is the radioactive isotope used in radiocarbon dating and radio labelling. The average atomic mass is a)12.5 b)12.1 c)6.05 d)6.1? defined & explained in the simplest way possible. Where Carbon 12 has six protons and six neutrons, Carbon 14 has six protons and eight neutrons. 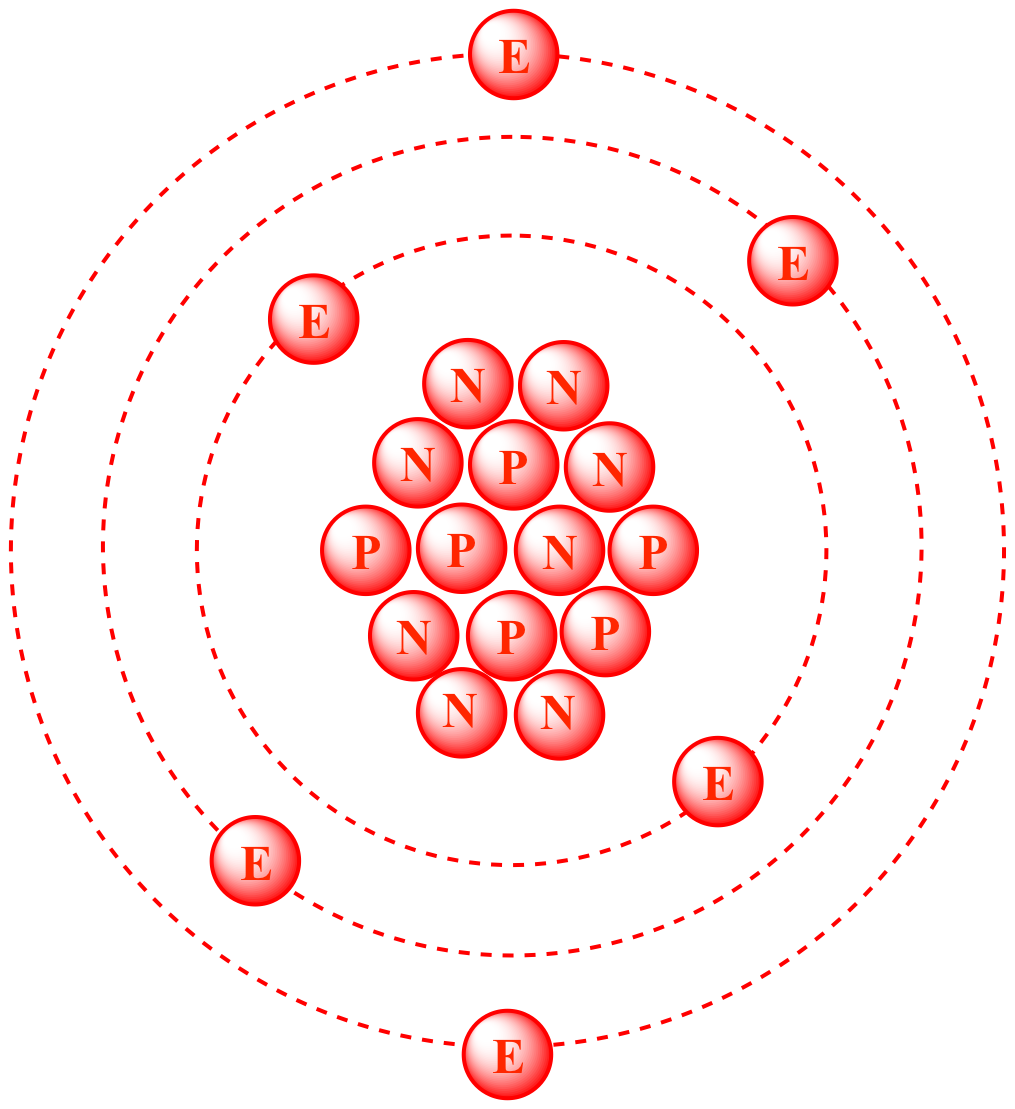
Here you can find the meaning of A sample of carbon that contains 95% by mole carbon-12 and 5% by mole carbon-14 isotopes. About 350 isotopes occur naturally on Earth, and another 1500 have been produced artificially. Question: Carbon-14 has the same atomic number and atomic mass as carbon-12 atomic number and thus number of neutrons as carbon-13 number of protons but more neutrons than carbon-12 O Oatomic mass as both carbon-12 and carbon-13 Submit Request Answer. When an organism dies, it stops taking in carbon-14, so the ratio of carbon-14 to carbon-12 in its remains, such as fossilized bones, will decline as carbon-14 decays gradually to nitrogen-14 2 ^2 2 squared. we can write carbon-12, carbon-13, and carbon-14. As animals eat the plants, or eat other animals that ate plants, the concentrations of carbon-14 in their bodies will also match the atmospheric concentration. As plants pull carbon dioxide from the air to make sugars, the relative amount of carbon-14 in their tissues will be equal to the concentration of carbon-14 in the atmosphere. These forms of carbon are found in the atmosphere in relatively constant proportions, with carbon-12 as the major form at about 99%, carbon-13 as a minor form at about 1%, and carbon-14 present only in tiny amounts 1 ^1 1 start superscript, 1, end superscript. For example, carbon is normally present in the atmosphere in the form of gases like carbon dioxide, and it exists in three isotopic forms: carbon-12 and carbon-13, which are stable, and carbon-14, which is radioactive.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
